Investigation into the mechanical stability of mechanophores by AFM-based single-molecule force spectroscopy
T. Carabin, 1 D. Sluysmans,1 G. De Bo 2 and A-S. Duwez 1
1 UR Molecular Systems, University of Liège. 2 School of Chemistry, University of Manchester
e-mail : t.carabin@uliege.be
Mechanical activation of chemical bonds is an ubiquitous process in nature. It governs many physiological activities, including enzyme activities, motion of molecular motors, division of cells, or muscle contraction. It is however a relatively unexplored source of energy in chemistry and material science. Exploiting the relationship between mechanical loads acting on a polymer and the tendency of its monomers to undergo chemical reactions offers new opportunities to create stress-responsive and energy-transducing materials. The inclusion of a mechanophore molecule, which contains scissile bonds that undergo predictable rupture under stress, in a polymer chain permits a precise control of this phenomenon1,2. However, the quantitative mechanical stability of these mechanophores and their mechanical reversibility are largely unknown. The mechanism by which the mechanical force can trigger a chemical reaction and its influence on the reversibility of the reaction is not clearly understood.
Here we have investigated the mechanical stability of several mechanophores based on Diels-Alder adducts incorporated in a polymer chain (PMA: poly(methyl acrylate)), using atomic force microscopy (AFM)-based single-molecule force spectroscopy. Our first standard single-molecule pulling experiments evidenced a characteristic mechanical response and allowed us to measure the rupture forces required to break open the adducts. We compared different regio- and stereoisomers in order to establish general relationships between the rupture force measured under mechanical load and molecular properties that govern the stability of these compounds.
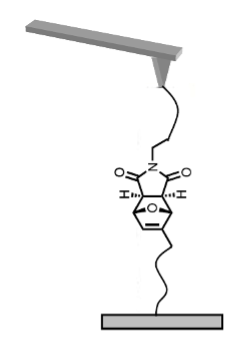
Figure 1: Schematic representation of the (AFM)-based single-molecule pulling experiment of a polymer chain comprising the mechanophore.
References
[1] Li, J., Nagamani, C. & Moore, J. S. Acc. Chem. Res. 2015, 48, 2181.
[2] Stevenson, R.; De Bo, G., J. Am. Chem. Soc. 2017, 139, 167
